HONG KONG, Jul 17, 2023 - (ACN Newswire) - In the treatment of type 2 diabetes mellitus (T2DM), glucokinase activator (GKA) is developed as anti-diabetic medicine with unique mechanism of action. After more than half a century since the discovery of glucokinase (GK), dorzagliatin, the world's first-in-class GKA drug, was approved for launch in China in 2022, and has received significant clinical attention. Several novel research advances on dorzagliatin were presented at the 2023 ADA Scientific Sessions, elaborating the mechanisms and benefits of GKA in improving glucose homeostasis. Let's check it out!
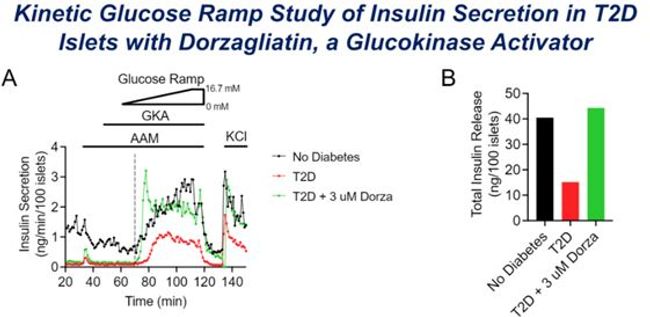
Figure1: Dorzagliatin restores GSIS threshold and overall insulin secretion
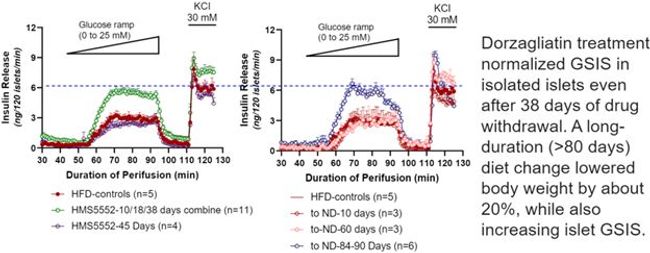
Figure 2: Dorzagliatin and diet change repairs GSIS function
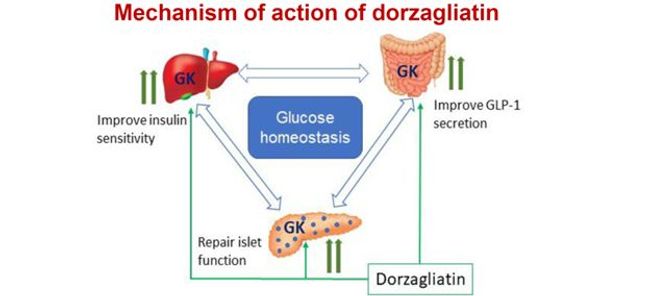
Figure 3: Mechanism of action of dorzagliatin
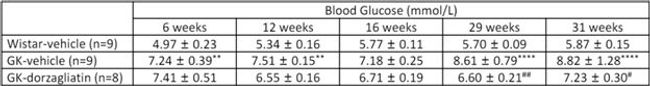
Dorzagliatin prevents hyperglycemia in GK rats
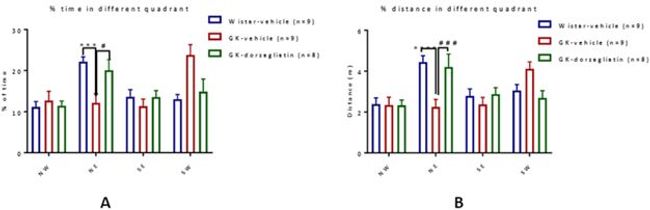
Figure 4: Dorzagliatin prevents memory impairment in GK rats
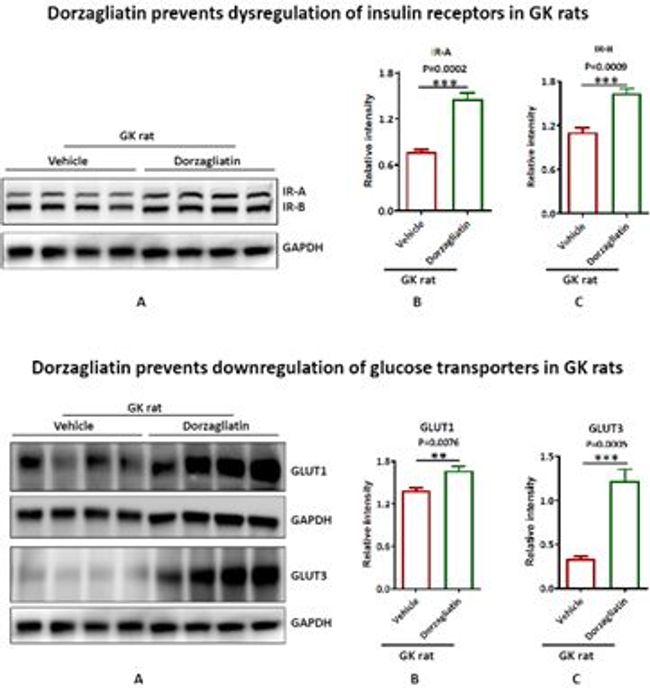
Figure 5: Dorzagliatin prevents dysregulation of insulin receptors in GK rats
293-LB
Dorzagliatin, a Glucokinase Activator, Restores Glucose-Sensing in Type 2 Diabetic Human Islets
Background
As a glucose sensor, GK plays a unique role in regulating glucose homeostasis. Stimulating GK improves the ability of pancreatic B-cells to detect the changes of glucose levels and release insulin accordingly known as glucose-stimulated insulin secretion (GSIS). GK is considered a drug target for T2DM due to its defects in those patients. Only a small number of GKA compounds, however, have so far entered clinical trials and most of them failed to advance to the market. For instance, MK-0941 induced hypoglycemia and other severe adverse events in clinical trials that led to the termination of clinical development. In contrast, Dorzagliatin, the first GKA approved for the treatment of T2DM, demonstrated a sustained reduction in HbA1c with no severe adverse events and low incidence of hypoglycemia (<1% in phase III trials). This study aims to compare the effects of dorzagliatin and other GKAs (including MK-0941) on the kinetics of the GK enzyme and the function of human islets from T2DM donors.
Methods:
Through the Integrated Islet Distribution Program, islets from 8 T2DM donors and 38 non-diabetic controls were tested for insulin secretion. To evaluate GSIS, glucose steps and glucose ramp perifusions protocol were used. The binding of GK-GK activator was studied via protein docking and molecular dynamics analysis.
Results:
In contrast to MK-0941, which powerfully activates GK enzyme even at very low glucose concentrations, dorzagliatin's activation of GK is glucose concentration-dependent. Molecular dynamics analysis demonstrates that dorzagliatin bonds to GK with less affinity than MK-0941. T2D islets exhibited a right-shifted glucose threshold for GSIS and/or a reduction in the maximal rate of GSIS (Vmax) in response to high glucose and IBMX, and dorzagliatin restores both the threshold and Vmax. In contrast, MK-0941 induces a rapid and transient insulin surge at extremely low glucose concentrations but has a diminished response at high glucose concentrations. The glucose dose-dependency of GK activation by dorzagliatin ensures its effects on amplifying the GSIS, while also limits the risk for hypoglycemia.
Figure1: Dorzagliatin restores GSIS threshold and overall insulin secretion
https://photos.acnnewswire.com/tr:n-650/20230717.GKA1.jpg
Conclusion:
In isolated T2DM islets, dorzagliatin restores both the normal glucose threshold and Vmax and has a greater dose range than MK-0941. These findings could explain why MK-0941 induces hypoglycemia and loses efficacy over time, while dorzagliatin improves blood glucose in T2DM patients without causing substantial adverse effects. Clinical success of dorzagliatin is facilitated by its unique effect on activating GK enzyme, which restores glucose sensitivity in islet B-cells of T2DM and thus normalizes GSIS.
297-LB
Dorzagliatin, a Glucokinase Activator, Repairs Defective Islet Function in High-Fat Diet-Induced Diabetic Mice
The purpose of this study is to examine the mechanisms of dorzagliatin-induced diabetes remission by evaluating the changes in islet function over time after a period of administering dorzagliatin in high-fat diet (HFD) induced obesity and diabetes mouse models.
Methods
Diabetes mice were generated after 5-7 months of HFD, then received dorzagliatin or vehicle controls via oral gavage once a day for 19 days (8 days of 30 mg/Kg and 11 days of 15 mg/Kg). After drug withdrawal, one group of mice remained in HFD and another group changed to a normal diet. The islets were then isolated at different time points after drug withdrawal. The glucose ramp-stimulated insulin secretion (GSIS) was evaluated in islet perfusion. Gene expression was measured by qPCR in islets and by RNA-seq in the liver.
Results
Compared to normal mouse islets, HFD-induced diabetic islets in vehicle groups have impaired GSIS with about 40% reduction in maximum insulin secretion. In contrast, GSIS was fully recovered to the levels of normal islets by dorzagliatin and was maintained in 10, 18 and 38 days after dorzagliatin withdrawal even the mice were still on HFD, however, GSIS returned to defective levels after 45 days of drug discontinuation. In addition, islet GSIS can be fully recovered after 80 days of diet change with 20% of body weight reduction. The study shows HFD-induced diabetic mice also have impaired liver glycogen metabolism. Dorzagliatin improved liver glycogen metabolism, likely by restoring islet function and liver insulin sensitivity. Dorzagliatin intervention, or dietary control and body weight reduction restore the key enzyme expression level back to normal.
Figure 2: Dorzagliatin and diet change repairs GSIS function
https://photos.acnnewswire.com/tr:n-650/20230717.GKA2.jpg
In conclusion, the defective GSIS in the islets of high-fat diet induced diabetic mice is reversible and can be entirely restored by dorzagliatin even after a prolonged period of drug withdrawal while still on a high-fat diet. The mechanisms are presumably the result of an increase in glucokinase gene expression caused by a brief dorzagliatin treatment. Dorzagliatin stimulates glucokinase and enhances tissue function in regions expressing GCK. This includes enhanced GLP-1 secretion, restored islet function, and increased liver insulin sensitivity, and these disease-modifying effects may lead to diabetes remission.
Figure 3: Mechanism of action of dorzagliatin
https://photos.acnnewswire.com/tr:n-650/20230717.GKA3.jpg
Figure 3: Dorzagliatin improves glucose sensitivity in pancreatic islets, intestinal endocrine cells and insulin sensitivity in the liver.
247-LB
Link between hyperglycemia and cognitive deficits in T2DM animal model-Dorzagliatin prevents hyperglycemia and cognitive deficits
Background and study objective
The Goto-Kakizaki (GK) rat is a model of spontaneously developing non-obese T2DM. GK rats develop insulin resistance, diminished pancreatic islet function, and hyperglycemia occurs as they age. Studies have shown that GK rats experience cognitive impairment concurrent with hyperglycemia, but the mechanisms are unclear. The purpose of this investigation is to study the potential link between hyperglycemia and cognitive impairment in this T2DM model and to determine whether dorzagliatin could prevent hyperglycemic cognitive abnormalities in GK rats.
Methods:
There were 3 groups of test subjects: male GK rats with placebo (n=9), male GK rats treated with dorzagliatin (n=8), and normal Wistar rats with placebo (n=9). At 6 weeks of age, oral administration of dorzagliatin or placebo began, and fasting blood glucose (FBG) and memory abilities were assessed.
Results
01 Blood Glucose Levels
Starting at 6 weeks of age, the FBG in the GK-vehicle group was significantly higher than in the Wistar-vehicle group, and there was progressive elevation of fasting hyperglycemia in GK-vehicle rats; however, long-term dorzagliatin treatment prevented FBG elevation.
Dorzagliatin prevents hyperglycemia in GK rats
https://photos.acnnewswire.com/tr:n-650/20230717.GKA4.jpg
02
Cognitive function
In the spatial probe test of Morris water maze (MWM) at 33 weeks of age, the GK-vehicle rats spent less time in the target quadrant and took more time to reach the location of the formerly hidden platform than the Wistar-vehicle rats, indicating that GK-vehicle rats showed spatial memory deficits, while the dorzagliatin treat GK rats showed similar performance to the normal rats.
Figure 4: Dorzagliatin prevents memory impairment in GK rats
https://photos.acnnewswire.com/tr:n-650/20230717.GKA5.jpg
03
Mechanism study
When GK rats administered a placebo, their brain expression of insulin receptors, glucose transporters, and synaptic proteins were all decreased, which led to the impairment of the insulin signaling pathway. These finding suggested that GK rat's brain has developed insulin resistance, dysregulated glucose uptake, impaired neuroplasticity, that caused neuropathological processes of memory defects.
Figure 5: Dorzagliatin prevents dysregulation of insulin receptors in GK rats
https://photos.acnnewswire.com/tr:n-650/20230717.GKA6.jpg
Conclusion
FBG rises with age in GK rats. Hyperglycemia is associated with cognitive impairment. Reduced glucose uptake in the brain is likely due to alterations in the insulin signaling pathway and decreased expression of essential synaptic proteins. A low dose of dorzagliatin administered continuously to juvenile GK rats maintains their glucose homeostasis and protects them from hyperglycemia, insulin resistance, and prevents cognitive impairment in adulthood.
Reference:
1. Doliba NM, et al. Dorzagliatin, a Glucokinase Activator, Restores Glucose-Sensing in Type 2 Diabetic Human Islets. Presented at ADA2023. 293-LB.
2. Han D, et al. Dorzagliatin, a Glucokinase Activator, Repairs Defective Islet Function in High-Fat Diet-Induced Diabetic Mice. Presented at ADA2023. 297-LB.
3. Ni J, et al. Link between Hyperglycemia and Cognitive Deficits in T2DM Animal Model-Dorzagliatin prevents hyperglycemia and cognitive dificits. Presented at ADA2023. 247-LB.
Copyright 2023 ACN Newswire. All rights reserved. www.acnnewswire.com