
TOKYO, May 28, 2021 - (JCN Newswire) - NEC Corporation (NEC; TSE: 6701) today announced the development of an AI technology for supporting doctors to detect neoplasia in Barrett's esophagus during endoscopic procedures. This is the world's first technology of its kind to comply with the requirements for CE mark labeling, a European safety, health and environmental protection standard (1), and is being released as WISE VISION Endoscopy, where it will soon be available in Europe to help doctors in the detection of Barrett's neoplasia (2).
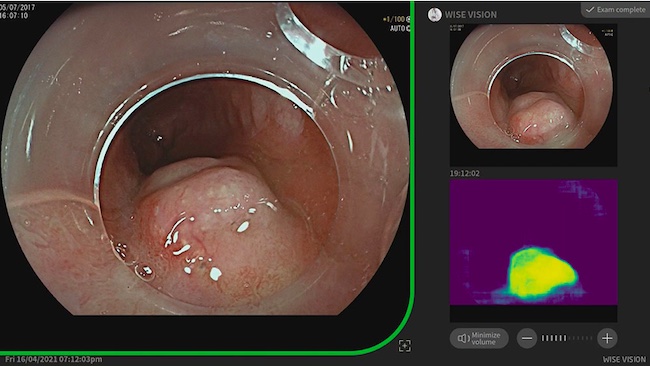
Provided by Professor Pradeep Bhandari: "Once the neoplasia is found, the system takes a still image and transfers it to the top right corner of a screen as a reference image for endoscopists. It also has a heat map which shows the area of the AI-predicted neoplasia."
This software is connected to endoscopy processors and automatically notifies users of potential Barrett's neoplasia from images captured during endoscopic procedures.
Barrett's esophagus is a pre-cancerous condition in which the inner lining of the esophagus is replaced by a lining that resembles that of the stomach. Worldwide prevalence of Barrett's esophagus is 1% of the entire population but much higher (7%) in patients with acid-reflux symptoms (3). Patients with Barrett's esophagus have 30-40 fold higher risk of developing cancer of the esophagus as compared to the normal population (4).
However, if this cancer can be detected at an early stage, then it can be removed through the endoscope and a patient can be cured. Patients with Barrett's esophagus have to undergo regular endoscopy (every 2-3 years) to detect early cancer, but this is still challenging as the early changes of cancer are very subtle and difficult to identify. As a result, current recommendations are to take multiple random biopsies throughout the length of Barrett's esophagus to identify any focus of early cancer (5, 6). However, random biopsies are expensive and time consuming and are only performed in accordance with official clinical guidelines by an estimated 30-51% of endoscopists (7, 8, 9, 10). Consequently, it has been pointed out that up to 40% of neoplasia may be missed (7) resulting in delayed cancer diagnosis and poor outcomes for the patients.
To help resolve this issue, NEC collaborated with Professor Pradeep Bhandari (Portsmouth, UK), Chair of the European Society of Gastrointestinal Endoscopy (ESGE) Research Committee, in development of this new technology to support doctors to detect Barrett's neoplasia during examination. WISE VISION Endoscopy has been trained with more than 1 million Barrett's esophagus endoscopy images, together with the knowledge of expert endoscopists, enabling it to meet the requirements for CE mark labeling. In developing this solution, NEC applied its face recognition technology that has been highly evaluated (11) by the National Institute of Standards and Technology (NIST) in the United States, and belongs to NEC's portfolio of cutting-edge AI technologies, "NEC the WISE."
On clinical evaluation, WISE VISION Endoscopy could find more than 90% of Barrett's neoplasia (12). WISE VISION Endoscopy is expected to significantly reduce the neoplasia miss rate during endoscopy and improve the outcome for patients with Barrett's esophagus all around the world.
The abstract applying this technology has received the Gut Award from the British Society of Gastroenterology (BSG), which was presented in the plenary session of the BSG Campus. It was also selected one of the best endoscopy abstracts in UEG Week Virtual 2020 held by United European Gastroenterology.
"I am delighted that NEC, as one of the world's leaders in AI technology, has entered the field of endoscopy and developed WISE VISION? Endoscopy to detect and help manage Gastrointestinal neoplasia. Barrett's neoplasia can be very flat and easily missed during endoscopy, despite taking multiple biopsies. I worked with NEC to develop the Barrett's neoplasia detection arm of WISE VISION? Endoscopy and am super impressed by the speed with which NEC has produced this as the World's first AI engine to obtain the CE mark for notifying Barrett's neoplasia.
"WISE VISION Endoscopy can notify very flat and subtle lesions and much faster than most endoscopists. Once the neoplasia is found, this system takes a still image and transfers it to the top right corner of a screen as a reference image for endoscopists. It doesn't stop here, as it also has a heat map which shows the area of the AI-predicted neoplasia. WISE VISION? Endoscopy is set to revolutionize the endoscopic detection and management of Barrett's neoplasia in the western world. I'm sure that clinicians involved in the management of Barrett's esophagus are looking forward to the dawn of this new (Wise) era in Endoscopy," said Professor Pradeep Bhandari, Chair of the ESGE Research Committee.
About WISE VISION Endoscopy
This software can be connected to endoscopes from three leading endoscope manufacturers (13). By simply connecting an existing endoscope to a monitor and terminal equipped with the software, users can start using it immediately. The system issues sound notifications when a candidate lesion has been located, and the type and volume of the notification can be customized at any time according to user preferences. Moreover, a highly visible user interface allows intuitive operation, enabling specialists to smoothly proceed with examinations.
Going forward, NEC aims to accelerate digital transformation in the medical field by providing safe and secure products and services, including AI, to promote a healthy, and sustainable society, where individuals can thrive from the utilization of advanced IT technologies.
(1) Source: NEC Corporation
(2) "WISE VISION" is a registered trademark of NEC Corporation in the United States of America, the European Union and the United Kingdom.
(3) Marques de S? I, Marcos P, Sharma P, Dinis-Ribeiro M. The global prevalence of Barrett's esophagus: A systematic review of the published literature. United European Gastroenterol J. 2020;8(9):1086-1105. doi:10.1177/2050640620939376
(4) Sharma P. Clinical practice. Barrett's esophagus. N Engl J Med. 2009 Dec 24;361(26):2548-56. doi: 10.1056/NEJMcp0902173. Erratum in: N Engl J Med. 2010 Apr 15;362(15):1450. PMID: 20032324.
(5) Weusten B, Bisschops R, Coron E, Dinis-Ribeiro M, Dumonceau JM, Esteban JM, Hassan C, Pech O, Repici A, Bergman J, di Pietro M. Endoscopic management of Barrett's esophagus: European Society of Gastrointestinal Endoscopy (ESGE) Position Statement. Endoscopy. 2017 Feb;49(2):191-198. doi: 10.1055/s-0042-122140. Epub 2017 Jan 25. PMID: 28122386.
(6) ASGE STANDARDS OF PRACTICE COMMITTEE, Qumseya B, Sultan S, Bain P, Jamil L, Jacobson B, Anandasabapathy S, Agrawal D, Buxbaum JL, Fishman DS, Gurudu SR, Jue TL, Kripalani S, Lee JK, Khashab MA, Naveed M, Thosani NC, Yang J, DeWitt J, Wani S; ASGE Standards of Practice Committee Chair. ASGE guideline on screening and surveillance of Barrett's esophagus. Gastrointest Endosc. 2019 Sep;90(3):335-359.e2. doi: 10.1016/j.gie.2019.05.012. PMID: 31439127.
(7) Kariv R, Plesec TP, Goldblum JR, et al. The Seattle protocol does not more reliably predict the detection of cancer at the time of esophagectomy than a less intensive surveillance protocol. Clinical gastroenterology and hepatology : the official clinical practice journal of the American Gastroenterological Association. 2009 Jun;7(6):653-8; quiz 06. PubMed PMID: 19264576. Epub 2009/03/07. eng.
(8) Shaheen NJ, Falk GW, Iyer PG, Gerson LB, American College of G. ACG Clinical Guideline: Diagnosis and Management of Barrett's Esophagus. Am J Gastroenterol. 2016;
(9) Spechler SJ, Sharma P, Souza RF, Inadomi JM, Shaheen NJ. American Gastroenterological Association technical review on the management of Barrett's esophagus. Gastroenterology. 2011 Mar;140(3):e18-52; quiz e13.
(10) Abrams JA, Kapel RC, Lindberg GM, Saboorian MH, Genta RM, Neugut AI, et al. Adherence to Biopsy Guidelines for Barrett's Esophagus Surveillance in the Community Setting in the United States. Clin Gastroenterol Hepatol [Internet]. 2009 Jul 1 [cited 2018 Nov 16];7(7):736?42.
(11) NEC Face Recognition Technology Ranks First in NIST Accuracy Testing
https://www.nec.com/en/press/201910/global_20191003_01.html
(12) Presented at ESGE Days 2021 held by European Society of Gastrointestinal Endoscopy
(13) Connectivity and operations have been confirmed with the following endoscopes.
Olympus EVIS LUCERA ELITE Video System Center CV-290
Olympus EVIS EXERA III Video System Center CV-190
FUJIFILM ELUXEO video processor VP-7000
PENTAX Medical OPTIVISTA EPK-i7010 video processor
PENTAX Medical OPTIVISTA EPK-i7000 video processor
For More Information
URL: https://www.nec.com/en/global/wisevision/?id=confirm
About NEC Corporation
NEC Corporation has established itself as a leader in the integration of IT and network technologies while promoting the brand statement of ?Orchestrating a brighter world.? NEC enables businesses and communities to adapt to rapid changes taking place in both society and the market as it provides for the social values of safety, security, fairness and efficiency to promote a more sustainable world where everyone has the chance to reach their full potential. For more information, visit NEC at https://www.nec.com.
Copyright 2021 JCN Newswire. All rights reserved. www.jcnnewswire.com